Molecular ‘blueprint’ illuminates how plants perceive light
March 31, 2022

GRAND RAPIDS, Mich. (March 30, 2022) — Plants rely on their ability to sense light for survival. But unlike animals, plants don’t have eyes full of photoreceptors to capture and convey messages from visual stimuli. Instead, plants are coated with a network of light-sensing photoreceptors that detect different wavelengths of light, allowing them to regulate their lifecycles and adjust to environmental conditions.
Now, Van Andel Institute and Washington University scientists have determined the molecular structure of one of these vital photoreceptors — a protein known as PhyB — revealing a wholly different structure than previously known. The findings, published today in Nature, may have implications for agricultural and “green” bioengineering practices.

“Photoreceptors, such as PhyB, help plants sense and respond to the world around them by influencing life-sustaining processes such as shade avoidance, seed germination, determination of flowering time, and development of chloroplasts, which convert light into usable energy,” said VAI Professor Huilin Li, Ph.D., co-corresponding author of the study. “Our new structure sheds light onto how PhyB works and has potential for a host of applications in agriculture, renewable energy and even in cellular imaging.”
Understanding the shape of PhyB is important because its structure directly impacts how PhyB interacts with other molecules to communicate shifts in light conditions and to help plants adapt by driving changes in gene expression. Previous research on PhyB provided only a truncated snapshot rather than a detailed rendering of the entire molecule.
To determine their near-atomic resolution image of PhyB, Li and study co-corresponding author Richard D. Vierstra, Ph.D., of Washington University, turned to one of the most studied plants on Earth — a humble weed called Arabidopsis thaliana. This small flowering plant is an ideal model for research because it reproduces quickly, is small and is easy to grow.
Using VAI’s high-powered cryo-electron microscope, or cryo-EM, the research team snapped nearly 1 million particle images of PhyB connected to its natural chromophore — a molecule that absorbs a certain color of light. They then narrowed the images down to 155,000, which they used to construct the full visualization of PhyB’s structure at a near-atomic level of 3.3 Ångstrom. Their work revealed a surprise: rather than the parallel structure described by earlier studies, they found a complicated 3D structure with both parallel and anti-parallel sections. The findings suggest that PhyB may amplify small changes in light-sensing chromophore molecules and drastically change its shape in response — a move that communicates the availability of light to the plant. 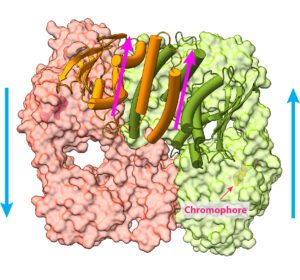
The discovery is the result of more than a decade of collaboration between Li and Vierstra, and revolutionizes what we know about PhyB and phytochromes, the family of receptors to which PhyB belongs. Until now, it was believed that PhyB and other phytochromes likely were similar to those used by single-celled organisms, such as certain bacteria. Today’s findings upend that theory and lay the foundations for further studies into the intricate details of PhyB and phytochrome function.
Hua Li, Ph.D., of VAI and Sethe Burgie, Ph.D., of Washington University are co-first authors of the study. Zachary T.K. Gannam, Ph.D., of Washington University also is an author. Cryo-EM data were collected in collaboration with VAI’s Cryo-EM Core and the David Van Andel Advanced Cryo-Electron Microscopy Suite.
Research reported in this publication was supported by Van Andel Institute (Li), Washington University (Vierstra) and the National Institute of General Medical Sciences of the National Institutes of Health under award nos. R01GM127892 (Vierstra) and R35GM131754 (Li). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
